Scroll to:
Orthostatic hypertension in cardiovascular risk stratification in hypertensive patients
https://doi.org/10.15829/560-4071-2021-4221
Abstract
Hypertension (HTN) is an important modifiable risk factor for cardiovascular disease associated with poor outcomes and high health care costs. The assessment of cardiovascular risk (CVR) according to the current ESC/ESH guidelines for the treatment of hypertensive patients presents a number of difficulties and initiates the search for new diagnostic methods that contribute to understanding the patient's phenotype, personalizing diagnostic and treatment tactics, and improving the outcomes of hypertensive patients. Regulatory mechanisms involved in the body's orthostatic response, such as activation of the sympathetic nervous system, catecholamine production, endothelial function, significantly contributes to maintaining blood pressure levels. Their violation plays an active role in hypertension development, which allows considering orthostatic HTN in a hypertensive patient as a marker of CVR stratification. The article discusses the diagnostic criteria for orthostatic HTN, its pathophysiological mechanisms and possible use as a marker of CVR stratification.
For citations:
Gubareva E.Yu., Fatenkov O.V., Gubareva I.V., Klimenko D.A., Shvan L.Yu., Limareva L.V. Orthostatic hypertension in cardiovascular risk stratification in hypertensive patients. Russian Journal of Cardiology. 2021;26(1S):4221. (In Russ.) https://doi.org/10.15829/560-4071-2021-4221
Hypertension (HTN) is the most important modifiable risk factor (RF) for cardiovascular diseases and remains one of the most significant medical and social problems in the world [1-3]. Annual mortality and disability, associated with increased blood pressure (BP), confirm a direct relationship between BP and cardiovascular risk (CVR) [1][4]. In addition, the incidence of HTN among young people increases, but this group often escapes the attention of physicians, which leads to a long absence of therapy, asymptomatic target organ damage and an increased risk of cardiovascular events (haemorrhagic and ischemic stroke, myocardial infarction, sudden death, heart failure and peripheral arterial disease), as well as end-stage renal failure [1][3].
Orthostatic test (OT) is an easy-to-perform and available method for assessing the cardiovascular system and its autonomic regulation, performed on an outpatient basis by an any physician and does not require additional equipment [3].
The change by the patient of body position from horizontal to the vertical one causes multidirectional changes in the hydrostatic pressure of cardiovascular system relative to some hydrostatic indifference point located several centimetres below the diaphragm level. The action of gravity shifts the intravascular volume from the chest (~400-1000 ml) into the splanchnic circulation and limb veins, complicating venous return and reducing the volume of circulating blood: most of the changes occur ~ in the first 10 seconds of orthostasis. The change in blood flow leads to a decrease in right ventricular filling and a decrease in cardiac output (CO), as a result of which there is a transient BP decrease and heart rate increase, which stimulates the baroreceptor areas and the central part of the autonomic nervous system (ANS), resulting in a decrease in vagal tone and increase in sympathetic tone. This reflex causes compensatory vasoconstriction of resistance and capacitance vessels in the visceral, musculocutaneous and renal vascular systems, an increase in peripheral vascular resistance and stroke volume, providing venous return, BP maintenance and organ perfusion [5-7]. Systemic vasoconstriction is a key factor in maintaining upright BP, more than an increase in heart rate. In a healthy person, orthostatic stabilization is achieved within 60 seconds and less.
The inability of regulatory mechanisms to adequately compensate for stress causes orthostatic intolerance, a type of which is orthostatic hypertension (OH). Since the regulatory mechanisms of the orthostatic response make a significant contribution to maintaining the blood pressure level, and their violation plays an active role in HTN development, this allows us to consider the presence of OH as a marker of CVR stratification [3].
Definition and diagnostic criteria for OH
The term ‘OH’, described as elevated blood pressure in an upright position and used by the medical community since 1940, is a condition rarely assessed by physical examination or regarded as an unexpected and illogical outcome of OT [5, 6].
The diagnostic criteria for OH are not defined [5][6], and the number of related studies is rather low: the term ‘OH’ is not included and is not defined by the current guidelines for HTN [1][8]. The proposed diagnostic criteria for OH were structured in the paper by Jordan J, et al. (2020) in a table (Table 1) [5]. Table 1 [5] includes studies [9-24], which are the first in the literature to suggest or apply specific definitions of OH, but none of them were based on normative data or CVR assessment. Most of them [10-16][20-24] used the absolute difference in systolic BP (SBP) and/or diastolic (DBP) in the horizontal position and orthostasis as a diagnostic criterion. The other studies [9][17][22][24] defined OH as the transition of normal BP in a horizontal position to increased BP in orthostasis, which depended on HTN definition [5].
Table 1
Diagnostic criteria for OH first published by Jordan J, et al. (2020) [5]
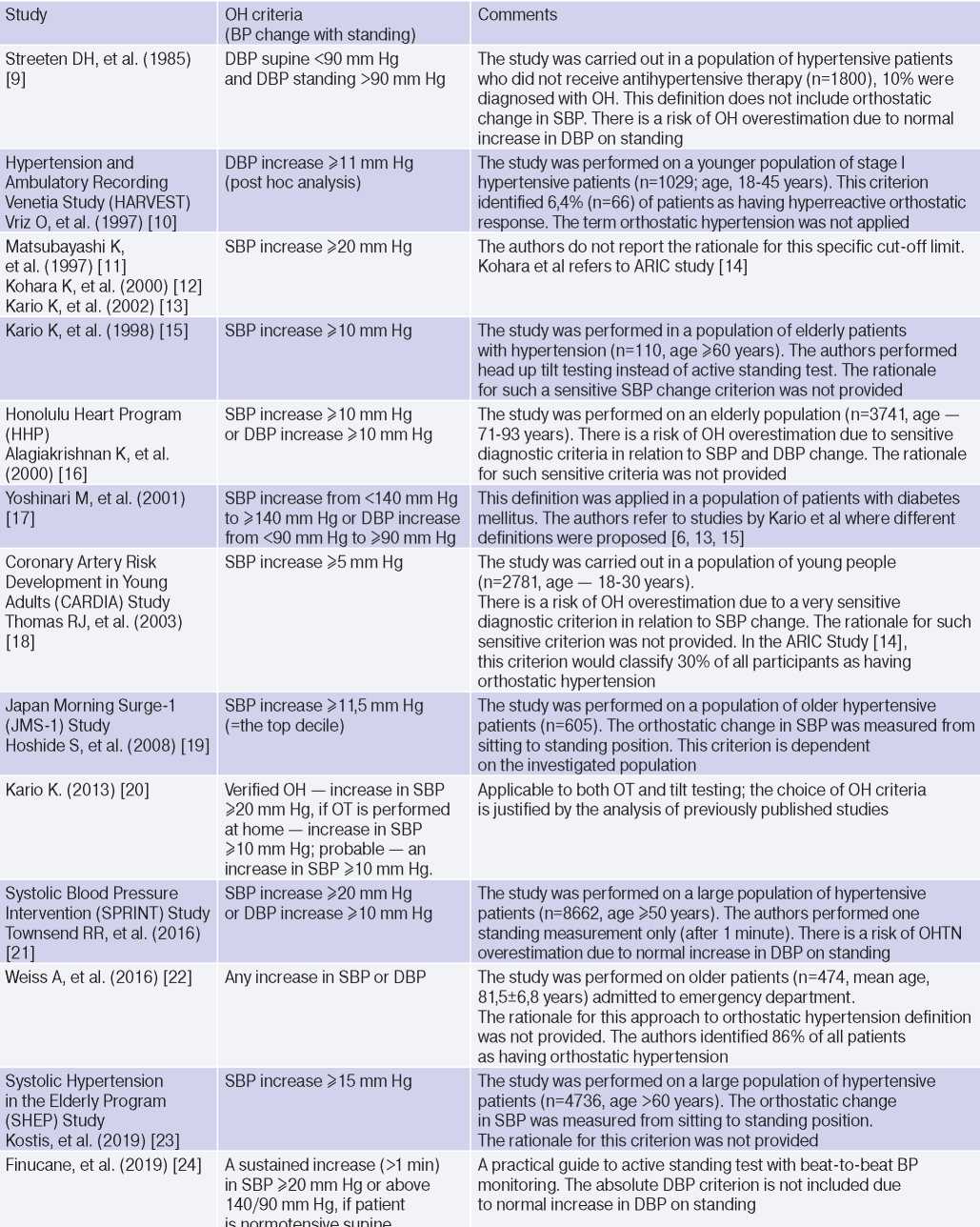
Abbreviations: HTN — hypertension, BP — blood pressure, DBP — diastolic blood pressure, SBP — systolic blood pressure, OH — orthostatic hypertension, OT — orthostatic test.
The use of increased DBP in orthostasis as a diagnostic criterion is less reliable, since in an upright position due to peripheral vasoconstriction and decreased stroke volume, an increase in DBP by 5-10 mm Hg is physiological [5][25]. Jordan J, et al. (2020) [5] consider an important and rarely discussed aspect of a patient’s BP change during OT: when should BP rise in orthostasis and how long should it remain elevated to confirm OH. In 2019, Finucane C, et al. proposed 2 diagnostic criteria for OH: a stable increase (>1 min) in SBP ≥20 mm Hg or >140 and 90 mm Hg, if the patient has normal BP in the horizontal position [5][24].
Pathogenetic mechanisms of OH
The pathogenetic mechanisms of OH are not fully understood [5][6][25]. The conventional explanation is the initial increased sympathetic activity and excessive sympathetic response caused by a decrease in CO as a result of orthostasis [6][9][13][20][25][26]. In addition, the pathogenetic mechanism of OH (Figure 1, first published in the paper by Magkas N, et al., 2019) may be caused by ANS dysfunction (impaired sensitivity of baroreceptors and/or baroreflex inability to adapt to “normal” BP), α-adrenergic vascular hyperactivity, increased norepinephrine concentration, renin-angiotensinaldosterone system (RAAS) activation and increased secretion of vasopressin [6][13][20][25][27-29]. Factors associated with OH development include arterial stiffness and remodelling of small arteries, excessive venous pooling [5][6][9][20][30][31].
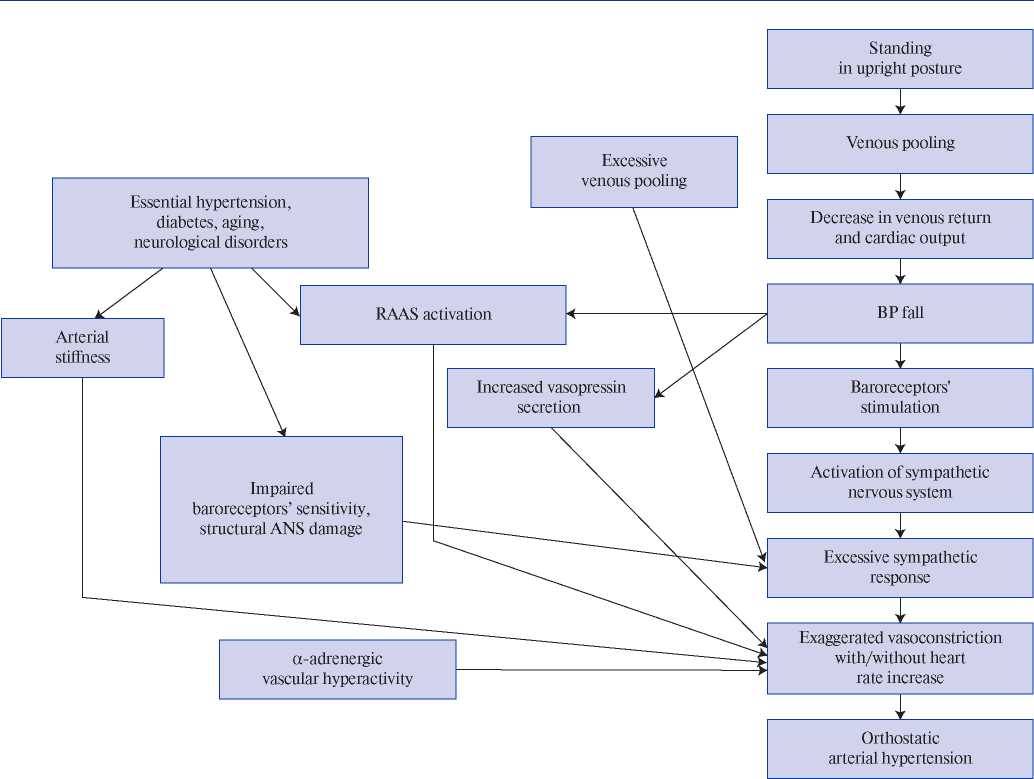
Figure 1. Mechanisms proposed to underlie pathogenesis of orthostatic hypertension; the figure first published in the paper by Magkas N., et al. (2019) [6].
Hypertension, along with aging, diabetes and neurological disorders, conditions associated with ANS degeneration and sympathetic activation, is considered as a precipitating factor of OH [12][18][20][32]. Structural and/or functional disorders of the renal vascular system, as in HTN, can also determine the pathogenetic mechanisms of OH. A decrease in renal blood flow activates the RAAS, further causing vasoconstriction, water and salt retention, and BP increase (renovascular hypertension, nephroptosis) [5][6][30][33][34]. Thus, it is obvious that OH and HTN have common pathogenetic mechanisms, but it remains unclear whether OH is a cause, effect or phenotype of HTN [6]?
Other rare causes of OH include vascular myelopathy, norepinephrine transporter deficiency, baroreflex failure, mutation of the gene encoding phosphodiesterase 3A, pheochromocytoma, mast cell activation syndrome, and posture disorders (Table 2) [5][6][25][35]. In children, endothelial damage and the related decrease in plasma levels of nitric oxide and nitric oxide synthase, vitamin D deficiency, as well as its participation in ANS and RAAS regulation are also considered pathogenetic mechanisms of OH [25][29][36].
Table 2
Conditions determining the pathogenetic mechanisms of OH

Epidemiology of OH and its potential use in CVR stratification for hypertensive patients
The absence of generally accepted diagnostic criteria for OH complicates the comparison of research results: in studies defining OH as an increase in SBP ≥20 mm Hg, the prevalence of OH varied from 1,1% in young patient population to 28% in elderly one [6, 25, 37, 38]. In studies using lower SBP elevation levels as diagnostic criteria, the prevalence of OH was expected to be higher.
OH is a pathological body response to orthostasis, but the consequences of its diagnosis in terms of CVR stratification are far from clear [3][5-7][13][15-19][21][32][38][46].
OH is interconnected with all components of the cardiovascular continuum in HTN: RFs, Asymptomatic HTN-mediated organ damage, and associated conditions (Table 3).
Table 3
Clinical studies studying OH and hypertension
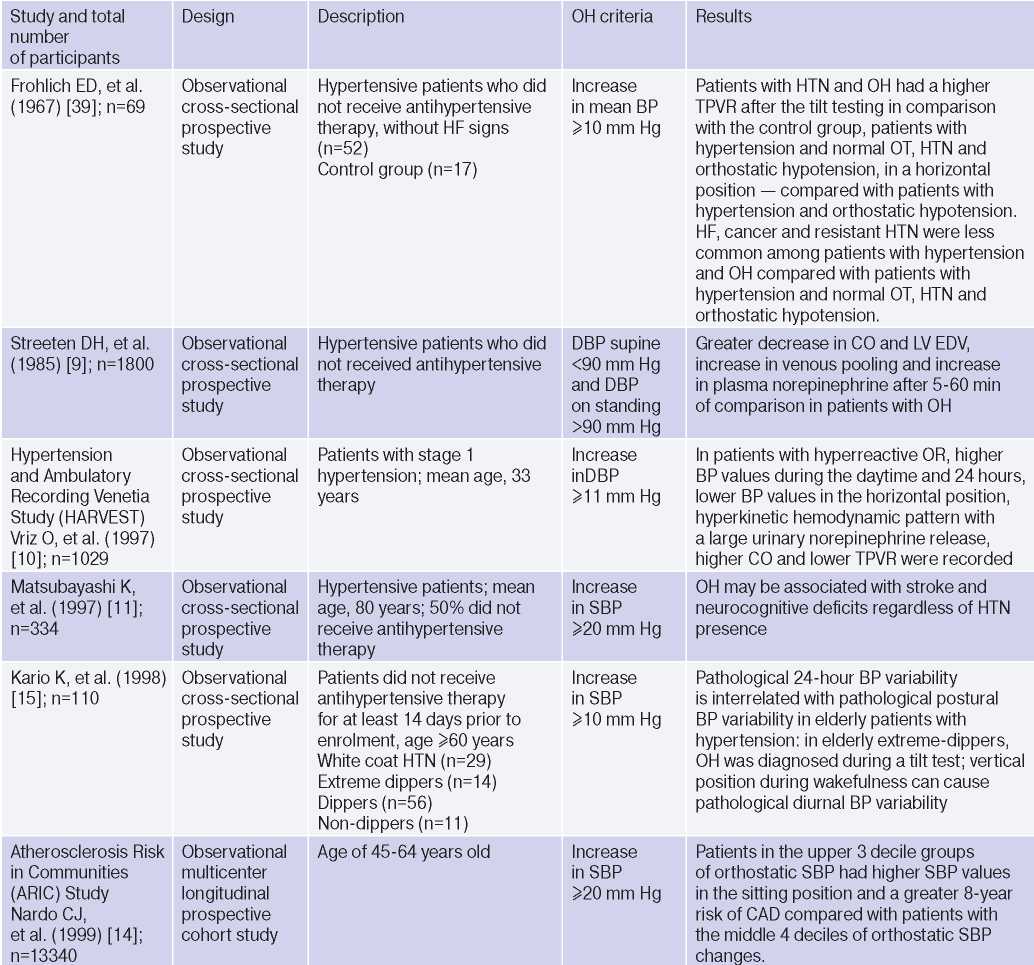
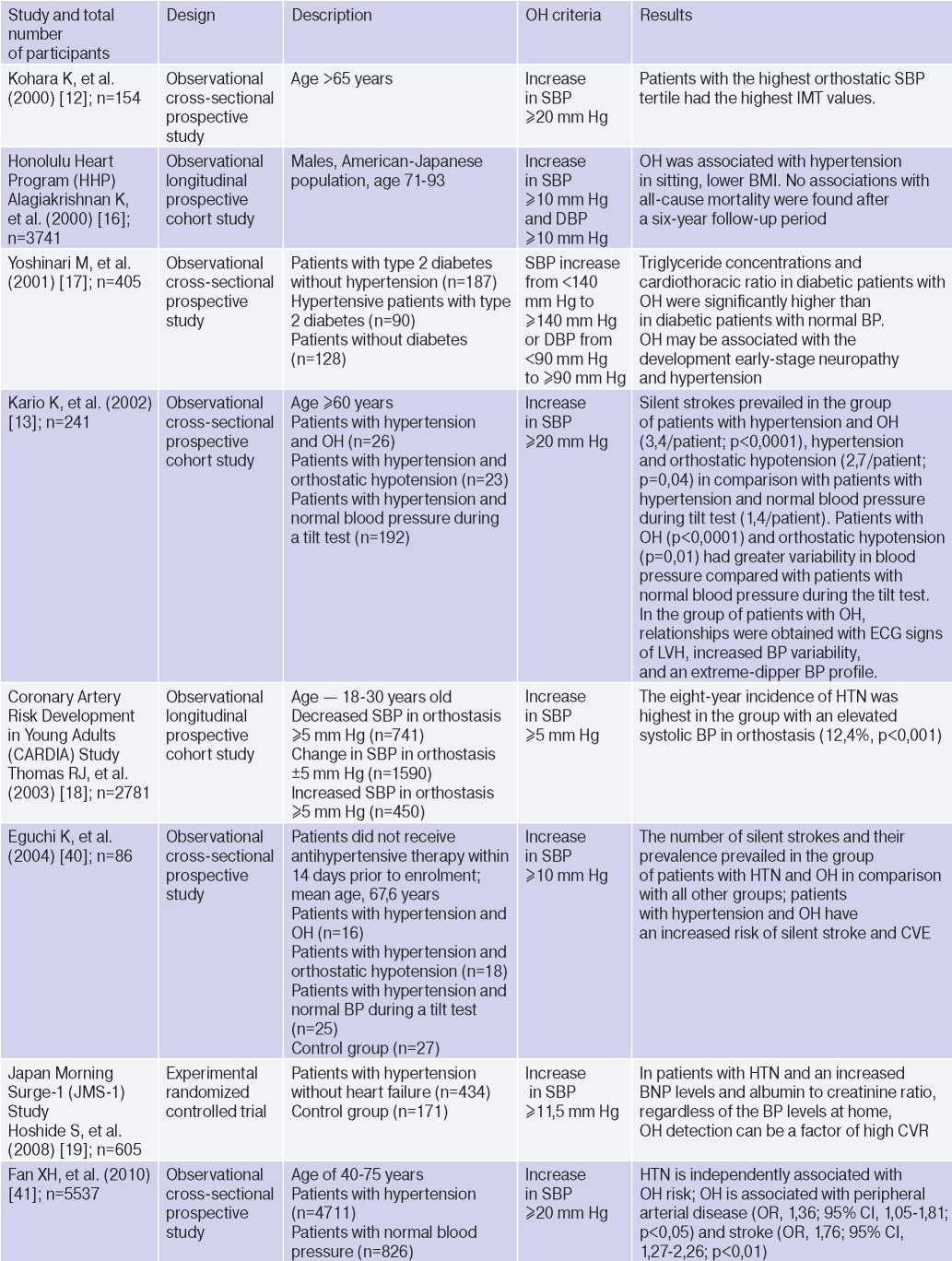
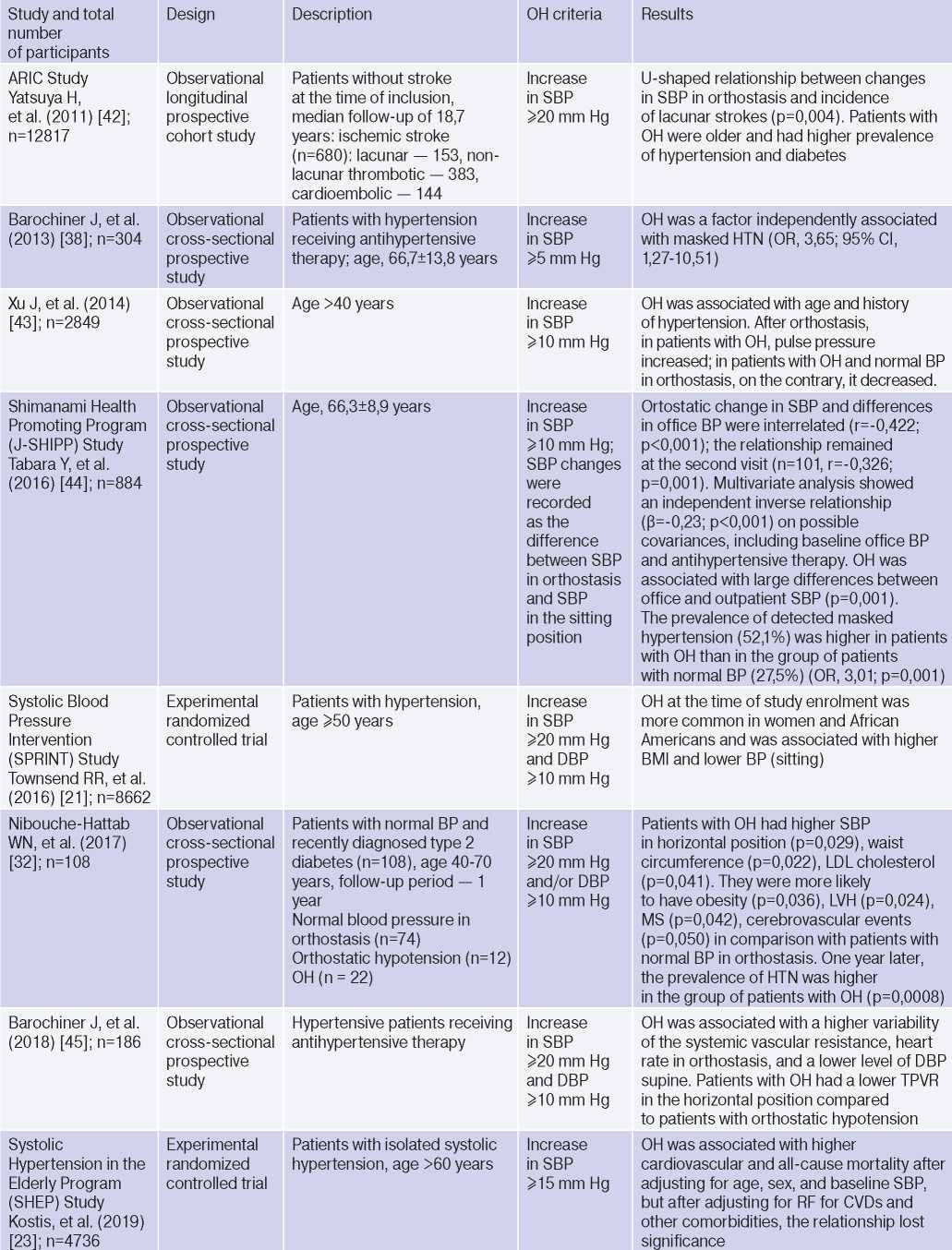
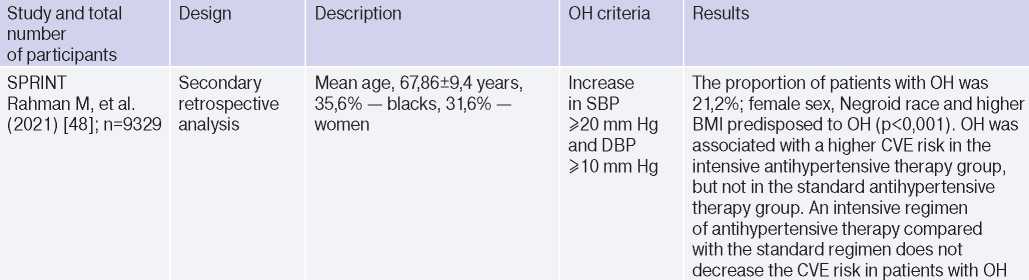
Abbreviations: HTN — hypertension, BP — blood pressure, LVH — left ventricular hypertrophy, DBP — diastolic blood pressure, CI — confidence interval, CAD — coronary artery disease, BMI — body mass index, LV EDV — left ventricular end diastolic volume, MS — metabolic syndrome, TPVR — total peripheral vascular resistance, OR — orthostatic reaction, OR — odds ratio, SBP — systolic blood pressure, CO — cardiac output, CVD — cardiovascular diseases, CVE — cardiovascular events, CVR — cardiovascular risk, IMT — intima– media thickness, RF — risk factors, LDL– low–density lipoprotein, HF — heart failure, BNP — brain natriuretic peptide.
The relationship between OH and HTN [6][10][13][16][18][21][36][37], hypertriglyceridemia [6][17][32], diabetes [6][17][42], body mass index, obesity and metabolic syndrome has been demonstrated [16][21][25][32][46], however, age, apparently, is decisive in OH pathogenesis [6][17][20][25][32][36][42][47]. The pathophysiological prerequisites and clinical characteristics of OH may differ depending on age and have different meanings: for a young patient, there is increased risk of HTN in the future [3][5][6][18][20][25][32], and in an elderly patient — independent CVR factor [13][20]. The relationship between OH and masked HTN was revealed [37][38].
OAS is interrelated not only with HTN, but also with BP changes during the day: the level of morning BP increase, BP variability and an excessive nocturnal BP decrease (Extrime-dipper profile), which are known markers of stroke [3][13][15][20]. Patients with extreme-dipper HTN have a higher prevalence of silent stroke diagnosed by magnetic resonance imaging compared with patients with dipper hypertension. In addition, patients with extremedipper hypertension have a greater risk of manifested stroke, and in case of the latter, have a worse prognosis. OH can be RF in hypertensive patients, because two-thirds of strokes in such extreme-dipper patients occur in the morning, when patients have a morning BP rise [15]. In 1997, Matsubayashi K, et al. [11] found a relationship of OH with stroke and neurocognitive deficits, independent of OH presence. The relationship between OH and the incidence of lacunar strokes was later confirmed by the ARIC study [42] and Nibouche-Hattab WN, et al. (2017) [32], while papers by Kario K, et al. (2002) [13], Eguchi K, et al. (2004) [40], Fan XH, et al. (2010) [41] revealed an increased risk of stroke in a cohort of patients with HTN and OH.
Cross-sectional studies have demonstrated the relationship between OH and HTN-mediated organ damage: an increase in natriuretic peptides and albumin-to-creatinine ratio [19], left ventricular hypertrophy [13][32], the intima-media thickness [12]; as well as associated clinical conditions: peripheral arterial disease [41] and coronary artery disease [14], which allows to consider the diagnosed OH as a factor of CVR stratification in HTN patients.
In 2019, Kostis, et al. [19], based on the Systolic Hypertension in the Elderly Program (SHEP) study, revealed an association of OH with higher cardiovascular and all-cause mortality after adjusting for age, sex, and baseline SBP. However, after adjusting for RFs of cardiovascular and other concomitant diseases, the relationship lost significance. In 2021, Rahman M, et al. published data from a retrospective analysis of the Systolic Blood Pressure Intervention Trial (SPRINT): in the intensive antihypertensive therapy group, OH was associated with a higher risk of cardiovascular outcomes [48].
Thus, although it is obvious that OH and HTN are interrelated conditions, the evidence base is small and there are no studies on the prognosis in patients with HTN and OH, and not in the general population of patients with OH. There are no criteria for the diagnosis of OH, approved by the medical community. Based on the foregoing, large-scale studies are needed to clarify the possibility of using diagnosed OH in hypertensive patients as a marker of CVR.
References
1. Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J Hypertens. 2018;36(10):1953-2041. doi:10.1097/HJH.0000000000001940. Erratum in: J Hypertens. 2019;37(1):226. doi:10.1097/HJH.0000000000002017.
2. NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19d million participants. Lancet. Lancet. 2017 Jan 7;389(10064):37-55. doi:10.1016/S0140-6736(16)31919-5. Erratum in: Lancet.;396(10255):886. doi:10.1016/S0140-6736(20)31972-3.
3. Sklyannaya EV. The role of orthostatic test in prognosis of arterial hypertension development in young adults. The Clinician. 2018;12(2):16-21. (In Russ.) doi:10.17650/1818-8338-2018-12-2-16-21.
4. Forouzanfar MH, Liu P, Roth GA, et al. Global Burden of Hypertension and Systolic Blood Pressure of at Least 110 to 115 mm Hg, 1990-2015. JAMA. 2017;317(2):165-82. doi:10.1001/jama.2016.19043. Erratum in: JAMA. 2017;317(6):648. doi:10.1001/jama.2017.0013.
5. Jordan J, Ricci F, Hoffmann F, et al. Orthostatic Hypertension: Critical Appraisal of an Overlooked Condition. Hypertension. 2020;75(5):1151-8. doi:10.1161/HYPERTENSIONAHA.120.14340.
6. Magkas N, Tsioufis C, Thomopoulos C, et al. Orthostatic hypertension: From pathophysiology to clinical applications and therapeutic considerations. J Clin Hypertens (Greenwich). 2019;21(3):426-33. doi:10.1111/jch.13491.
7. Gutkin M, Stewart JM. Orthostatic Circulatory Disorders: From Nosology to Nuts and Bolts, American Journal of Hypertension. 2016;29(9):1009-19. doi:10.1093/ajh/hpw023.
8. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/ APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):e13-e115. doi:10.1161/HYP.0000000000000065. Erratum in: Hypertension. 2018;71(6):e140-e144. doi:10.1161/HYP.0000000000000076.
9. Streeten DH, Auchincloss JH Jr, Anderson GH Jr, et al. Orthostatic hypertension. Pathogenetic studies. Hypertension. 1985;7:196-203. doi:10.1161/01.hyp.7.2.196.
10. Vriz O, Soon G, Lu H, et al. Does orthostatic testing have any role in the evaluation of the young subject with mild hypertension? An insight from the HARVEST study. Am J Hypertens. 1997;10(5 pt 1):546-51. doi:10.1016/s0895-7061(96)00489-x.
11. Matsubayashi K, Okumiya K, Wada T, et al. Postural dysregulation in systolic blood pressure is associated with worsened scoring on neurobehavioral function tests and leukoaraiosis in the older elderly living in a community. Stroke. 1997;28:2169-73. doi:10.1161/01.str.28.11.2169.
12. Kohara K, Tabara Y, Yamamoto Y, Miki T. Orthostatic hypertension: another orthostatic disorder to be aware of. J Am Geriatr Soc. 2000;48:1538-9. doi:10.1111/jgs.2000.48.11.1538.
13. Kario K, Eguchi K, Hoshide S, et al. U-curve relationship between orthostatic blood pressure change and silent cerebrovascular disease in elderly hypertensives: orthostatic hypertension as a new cardiovascular risk factor. J Am Coll Cardiol. 2002;40:133-41. doi:10.1016/s0735-1097(02)01923-x.
14. Nardo CJ, Chambless LE, Light KC, et al. Descriptive epidemiology of blood pressure response to change in body position. The ARIC study. Hypertension. 1999;33:1123-9. doi:10.1161/01.hyp.33.5.1123.
15. Kario K, Eguchi K, Nakagawa Y, et al. Relationship between extreme dippers and orthostatic hypertension in elderly hypertensive patients. Hypertension. 1998;31:77-82. doi:10.1161/01.hyp.31.1.77.
16. Alagiakrishnan K, Masaki K, Schatz I, et al. Postural hypertension in elderly men — the Honolulu Heart Program. Hawaii Med J. 2000;59:48-50.
17. Yoshinari M, Wakisaka M, Nakamura U, et al. Orthostatic hypertension in patients with type 2 diabetes. Diabetes Care. 2001;24:1783-6. doi:10.2337/diacare.24.10.1783.
18. Thomas RJ, Liu K, Jacobs DR Jr, et al. Positional change in blood pressure and 8-year risk of hypertension: the CARDIA Study. Mayo Clin Proc. 2003;78:951-8. doi:10.4065/78.8.951.
19. Hoshide S, Matsui Y, Shibasaki S, et al.; Japan Morning Surge-1 Study Group. Orthostatic hypertension detected by self-measured home blood pressure monitoring: a new cardiovascular risk factor for elderly hypertensives. Hypertens Res. 2008;31:1509-16. doi:10.1291/hypres.31.1509.
20. Kario K. Orthostatic hypertension — a new haemodynamic cardiovascular risk factor. Nat Rev Nephrol. 2013;9(12):726-38. doi:10.1038/nrneph.2013.224.
21. Townsend RR, Chang TI, Cohen DL, et al.; SPRINT Study Research Group. Orthostatic changes in systolic blood pressure among SPRINT participants at baseline. J Am Soc Hypertens. 2016;10:847-56. doi:10.1016/j.jash.2016.08.005.
22. Weiss A, Beloosesky Y, Grossman A, et al. The association between orthostatic hypertension and all-cause mortality in hospitalized elderly persons. J Geriatr Cardiol. 2016;13:239-43. doi:10.11909/j.issn.1671-5411.2016.03.004.
23. Kostis WJ, Sargsyan D, Mekkaoui C, et al. Association of orthostatic hypertension with mortality in the systolic hypertension in the elderly program. J Hum Hypertens. 2019;33:735-40. doi:10.1038/s41371-019-0180-4.
24. Finucane C, van Wijnen VK, Fan CW, et al. A practical guide to active stand testing and analysis using continuous beat-to-beat non-invasive blood pressure monitoring. Clin Auton Res. 2019;29:427-41. doi:10.1007/s10286-019-00606-y.
25. Hu Y, Jin H, Du J. Orthostatic Hypertension in Children: An Update. Front Pediatr. 2020;8:425. doi:10.3389/fped.2020.00425.
26. Lee H, Kim HA. Orthostatic hypertension: An underestimated cause of orthostatic intolerance. Clin Neurophysiol. 2016;127(4):2102-7. doi:10.1016/j.clinph.2015.12.017.
27. Buddineni JP, Chauhan L, Ahsan ST, Whaley-Connell A. An Emerging Role for Understanding Orthostatic Hyp'er'tension in the Cardiorenal Syndrome. Cardiorenal Med. 2011;1(2):113-22. doi:10.1159/000327141.
28. Zhao J, Yang J, Du S, et al. Changes of atrial natriuretic peptide and antidiuretic hormone in children with postural tachycardia syndrome and orthostatic hypertension: a case control study. Chin Med J (Engl). 2014;127(10):1853-7.
29. Sun X, Zou R, Luo X, et al. Changes in 25 hydroxyvitamin D level in school-aged children with orthostatic hypertension. Chin J Appl Clin Pediatr. 2018;33:32-5. doi:10.3760/cma.j.issn.2095-428X.2018.01.008.
30. Wijkman M, Lanne T, Ostgren CJ, Nystrom FH. Diastolic orthostatic hypertension and cardiovascular prognosis in type 2 diabetes: a prospective cohort study. Cardiovasc Diabetol. 2016;15:83. doi:10.1186/s12933-016-0399-0.
31. Hoshide S, Kario K, Eguchi K, et al. Altered aortic properties in elderly orthostatic hypertension. Hypertens Res. 2005;28(1):15-9. doi:10.1291/hypres.28.15.
32. Nibouche-Hattab WN, Lanasri N, Zeraoulia F, et al. Orthostatic hypertension in normotensive type 2 diabetics: What characteristics? Ann Cardiol Angeiol (Paris). 2017;66(3):159-64. doi:10.1016/j.ancard.2017.04.003.
33. Samadian F, Dalili N, Jamalian A. New insights into pathophysiology, diagnosis, and treatment of renovascular hypertension. Iran J Kidney Dis. 2017;11(2):79-89.
34. Schiefer J, Amthauer H, Genseke P, et al. Position-related renal perfusion disturbances as a possible under-estimated mechanism in patients with resistant hypertension: a case vignette. Int Urol Nephrol. 2017;49(10):1823-33. doi:10.1007/s11255-017-1656-1.
35. Tabara Y, Masaki M, Ikezoe T, et al. Small Degree of Lumbar Lordosis as an Overlooked Determinant for Orthostatic Increases in Blood Pressure in the Elderly: The Nagahama Study. Am J Hypertens. 2019;32(1):61-9. doi:10.1093/ajh/hpy137.
36. Zhao J, Du S, Yang J, et al. Changes in plasma nitric oxide and nitric oxide synthase activity in children with orthostatic hypertension. Chin Appl Clin Pediatr. 2014;29:971-3. doi:10.3760/j.issn.2095-428X.2014.13.005.
37. Wu JS, Yang YC, Lu FH, et al. Population-based study on the prevalence and correlates of orthostatic hypotension/hypertension and orthostatic dizziness. Hypertens Res. 2008;31(5):897-904. doi:10.1291/hypres.31.897.
38. Barochiner J, Cuffaro PE, Aparicio LS, et al. Predictors of masked hypertension among treated hypertensive patients: an interesting association with orthostatic hypertension. Am J Hypertens. 2013;26(7):872-8. doi:10.1093/ajh/hpt036.
39. Frohlich ED, Tarazi RC, Ulrych M, et al. Tilt test for investigating a neural component in hypertension. Its correlation with clinical characteristics. Circulation. 1967;36(3):387-93.
40. Eguchi K, Kario K, Hoshide S, et al. Greater change of orthostatic blood pressure is related to silent cerebral infarct and cardiac overload in hypertensive subjects. Hypertens Res. 2004;27(4):235-41. doi:10.1291/hypres.27.235.
41. Fan XH, Wang Y, Sun K, et al. Disorders of orthostatic blood pressure response are associated with cardiovascular disease and target organ damage in hypertensive patients. Am J Hypertens. 2010;23(8):829-37. doi:10.1038/ajh.2010.76.
42. Yatsuya H, Folsom AR, Alonso A, et al.; ARIC Study Investigators. Postural changes in blood pressure and incidence of ischemic stroke subtypes: the ARIC study. Hypertension. 2011;57(2):167-73. doi:10.1161/HYPERTENSIONAHA.110.161844.
43. Xu J, Zhou Y, Cao K, et al. Excessive pulse pressure response to standing in community population with orthostatic systolic hypertension. J Am Soc Hypertens. 2014;8(3):166-70. doi:10.1016/j.jash.2013.12.002.
44. Tabara Y, Igase M, Miki T, et al. Orthostatic hypertension as a predisposing factor for masked hypertension: the J-SHIPP study. Hypertens Res. 2016;39(9):664-9. doi:10.1038/hr.2016.43.
45. Barochiner J, Aparicio LS, Alfie J, et al. Hemodynamic characterization of hypertensive patients with an exaggerated orthostatic blood pressure variation. Clin Exp Hypertens. 2018;40(3):287-91. doi:10.1080/10641963.2017.1368539.
46. Hu Y, He B, Han Z, et al. Risk Factors for Orthostatic Hypertension in Children. J Pediatr. 2020;227:212-7.e1. doi:10.1016/j.jpeds.2020.07.030.
47. Kang M, Xu Y, Zou R, et al. Differences of age and gender in orthostatic hypertension-a single-center study. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(8):783-8. (In Chinese). doi:10.11817/j.issn.1672-7347.2016.08.002.
48. Rahman M, Pradhan N, Chen Z, et al. Orthostatic Hypertension and Intensive Blood Pressure Control; Post-Hoc Analyses of SPRINT. Hypertension. 2021;77(1):49-58. doi:10.1161/HYPERTENSIONAHA.120.15887.
About the Authors
E. Yu. GubarevaRussian Federation
Samara.
Competing Interests:
No
O. V. Fatenkov
Russian Federation
Samara.
Competing Interests:
No
I. V. Gubareva
Russian Federation
Samara.
Competing Interests:
No
D. A. Klimenko
Russian Federation
Samara.
Competing Interests:
No
L. Yu. Shvan
Russian Federation
Samara.
Competing Interests:
No
L. V. Limareva
Russian Federation
Samara.
Competing Interests:
No
Supplementary files
Review
For citations:
Gubareva E.Yu., Fatenkov O.V., Gubareva I.V., Klimenko D.A., Shvan L.Yu., Limareva L.V. Orthostatic hypertension in cardiovascular risk stratification in hypertensive patients. Russian Journal of Cardiology. 2021;26(1S):4221. (In Russ.) https://doi.org/10.15829/560-4071-2021-4221

















































